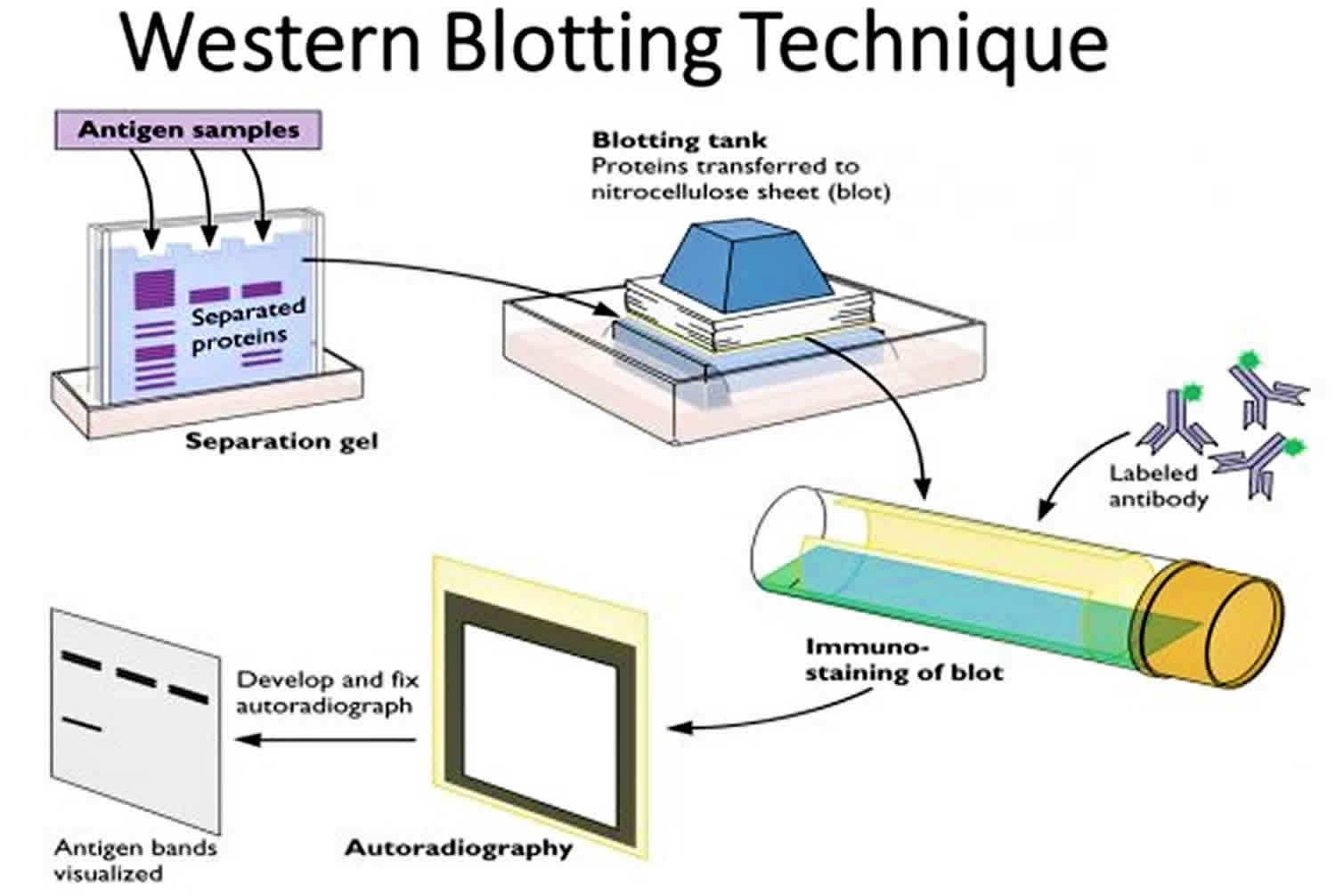
A very widespread discontinuous buffer system is the tris-glycine or "Laemmli" system that stacks at a pH of 6.8 and resolves at a pH of ~8.3-9.0. The "Laemmli" System: Creating The Environment For ElectrophoresisĪ buffer system with different pH values is applied in gel electrophoresis process. An appropriate amount of glycerol is added to increase density and accelerate the migration of sample solution. A bromophenol blue dye may be added to the protein solution to allow the experimenter to track the progress of the protein solution through the gel during the electrophoretic run. Heating the samples to at least 60☌ further promotes protein denaturation and depolymerization, helping SDS to bind and enabling the rod-shape formation and negative charge adherence. And the mixed samples are subsequently treated by related solution. The sample to be analyzed is mixed with SDS. Thus electrophoretic mobility of the SDS-protein subunit compound is based on molecular weight, eliminating the influence imposed by size and charge. And the length of major axis is in proportion to molecular weight of the protein subunit. Minor axis of all rods, the SDS-protein subunit compound are nearly the same, about 1.8nm. The electrostatic repulsion that is created by binding of SDS causes proteins to unfold into a rod-like shape thereby eliminating differences in shape as a factor for separation in the gel. This method is used in the fields of molecular biology, biochemistry, immunogenetics and other molecular biology disciplines. Western blotting analysis could detect target protein which is as low as 1ng due to high resolution of the gel electrophoresis and strong specificity and high sensitivity of the immunoassay. By analyzing location and intensity of the specific reaction, expression details of the target proteins in the given cells or tissue homogenate could be obtained. The separated proteins are then transferred or blotted onto a matrix (generally nitrocellulose or PVDF membrane), where they are stained with antibodies (used as a probe) specific to the target protein. 
to separate native proteins by 3-D structure or denatured proteins by the length of the polypeptide). It uses SDS-polyacrylamide gel electrophoresis (SDS-PAGE) to separate various proteins contained in the given sample (e.g. Western blotting (also called Protein Immunoblotting because an antibody is used to specifically detect its antigen) is a widely accepted analytical technique used to detect specific proteins in the given sample. What Is western blot? Western Blotting separates proteins by size and labels the protein of interest with an antibody
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
